Glucose Hypometabolism and p-Tau Reveal a Synergistic Mechanism in Alzheimer’s Disease
Date:2026-04-28
In Alzheimer’s disease (Alzheimer’s disease, AD), the abnormal accumulation of phosphorylated Tau protein (p-Tau) within neurons is closely associated with neurodegeneration. Meanwhile, reduced cerebral glucose metabolism has been widely recognized as an important clinical indicator for predicting cognitive decline. For a long time, these two hallmark pathological features—one resembling a “tangle” that entraps neurons and the other acting like a “switch” that cuts off energy supply—have been extensively studied. However, how they cooperate to drive AD progression has remained an important unresolved scientific question.
On April 22, 2026, the team led by Chengyu Zou at the Interdisciplinary Research Center on Biology and Chemistry, Shanghai Institute of Organic Chemistry, CAS, in collaboration with the team of Cong Liu and Professor Jochen Herms from Ludwig Maximilian University of Munich / German Center for Neurodegenerative Diseases, published a research article entitled “Glucose hypometabolism and hyperphosphorylated Tau synergistically drive neuronal necroptosis” in Neuron. This study, for the first time, elucidates the molecular mechanism by which reduced glucose metabolism and p-Tau synergistically drive AD progression through regulating the RIPK1-mediated necroptosis pathway.

Through a series of in vitro cellular experiments and in vivo animal model studies, the researchers demonstrated that glucose hypometabolism and p-Tau do not act independently; instead, they exert a synergistic effect to induce neuronal necroptosis and ultimately lead to neuronal loss. Under low-glucose conditions, abnormally accumulated p-Tau functions as a molecular scaffold that directly recruits RIPK1, a key kinase in the necroptosis pathway. Concurrently, the expression of the necroptosis checkpoint protein A20 is downregulated, weakening its inhibitory effect on this pathway and thereby releasing a critical brake on cell death. This coordinated regulatory mechanism promotes neuronal necroptosis.
At the intervention level, the study further showed that restoring A20 expression through supplementation with acetyl-L-carnitine (ALCAR), or disrupting the interaction between p-Tau and RIPK1 using a peptide derived from the intermediate domain of RIPK1, can significantly inhibit neuronal necroptosis in Tau transgenic mouse models. These interventions also alleviate brain atrophy and improve learning and memory deficits. Collectively, this work identifies a metabolism-driven necroptosis mechanism mediated by the p-Tau–RIPK1 axis, providing important insights into the functional link between brain glucose metabolism and Tau pathology, and offering new therapeutic strategies for AD.
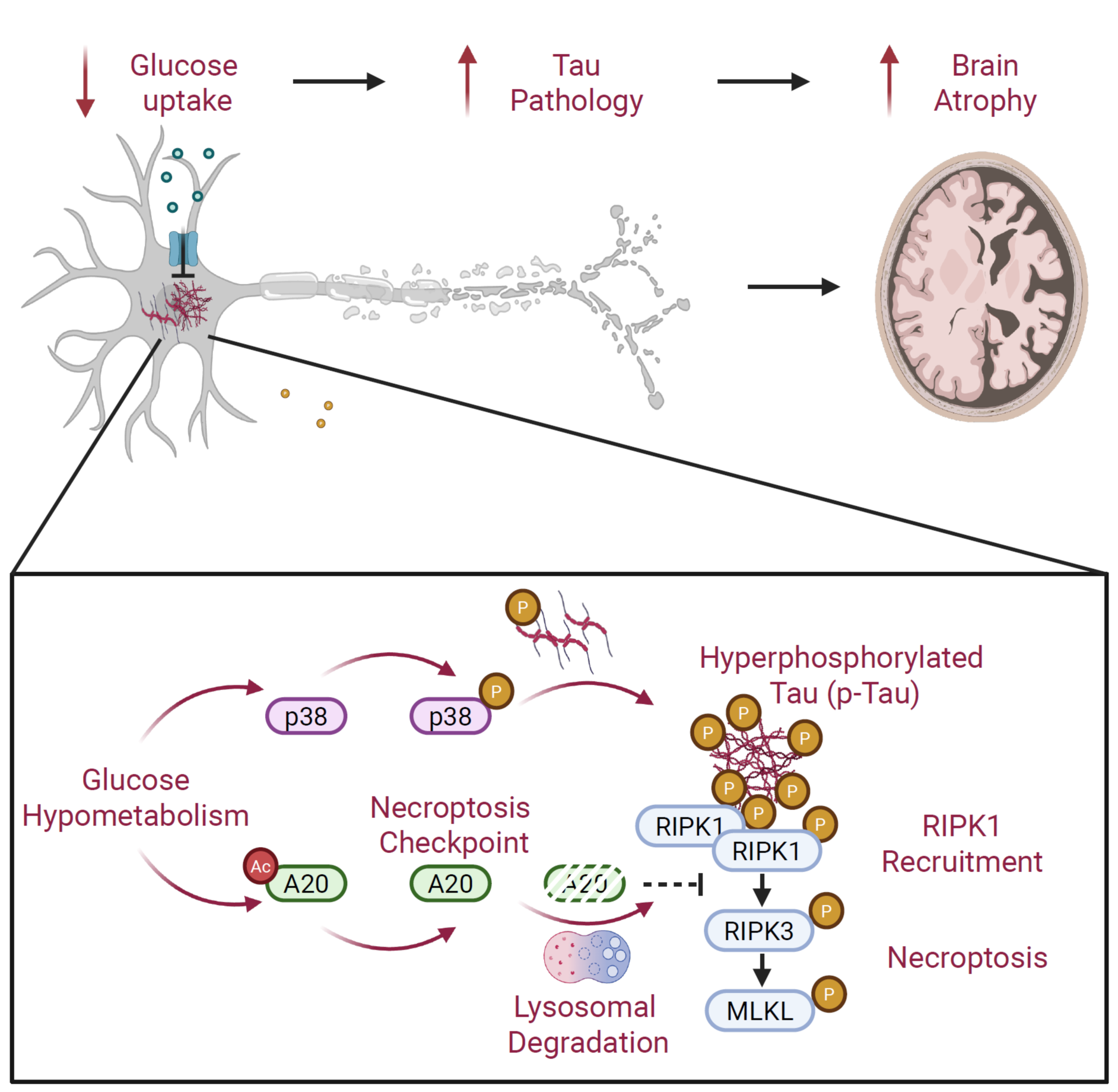
Figure 1. Schematic diagram illustrating the mechanism by which glucose hypometabolism and p-Tau synergistically drive neuronal necroptosis
Notably, this study reflects a century-spanning legacy of scientific discovery. Professor Jochen Herms’ Institute of Neuropathology at Ludwig Maximilian University of Munich is historically significant as a key site where Alois Alzheimer conducted his pioneering research. The term “Alzheimer’s disease” (Alzheimersche Krankheit) was first coined by Emil Kraepelin, a former director of the institute. On the other hand, necroptosis, the critical form of programmed cell death highlighted in this study, was first discovered and named in 2005 by Academician Junying Yuan, Director of the Center for Excellence in Molecular Cell Science. The convergence of a century-old disease discovery and a modern breakthrough in cell death research forms the conceptual foundation of this work. Building upon these two landmark “from 0 to 1” discoveries, the present study further advances our understanding of AD pathogenesis and proposes potential intervention strategies.
Article Link:https://www.cell.com/neuron/abstract/S0896-6273(26)00265-5
附件下载:
